Fear and Medical Side Effects
What if all it takes for medicine to hurt people is the belief that it hurts people?
This was a timed post. The way these work is that if it takes me more than an hour to complete the post, an applet that I made deletes everything I’ve written so far and I abandon the post. You can find my previous timed post here.
Many medical treatments, dietary choices, and physical activities seem to induce “nocebo effects”—harm through expectation, even when expectations aren’t met.
The nocebo is a variant of the placebo effect, but it’s far more real than placebo effects tend to be. Nocebo effects are insidious and pervasive, and they drive a lot of the online discussion surrounding topics like mold, fluoride, MSG, sugar, seed oils, and more. For example, people who self-identify as having non-celiac gluten sensitivity have side effects from the belief that they’ve consumed gluten more often than they have effects that can be ascribed to the actual consumption of gluten.
The impact of the nocebo effect on public health knowledge and practice is shockingly large. To make this clear, I’ll talk about the case of statins: very cheap, highly-effective medications that greatly reduce the risk of cardiovascular disease, extending lifespans by years for people who start them early enough.1
Despite their incredible efficacy, statins get a lot of hate, largely because of misperceptions about their side effects. Patients who run into issues with statins are deemed ‘statin intolerant’ and ideally shifted to alternative treatment options like intensive lifestyle interventions, ezetimibe, PCSK9 or ACL inhibitors, fibrates, or bile acid sequestrants. The portion of patients who actually become intolerant ranges from about 5% in trials to 17% in observational studies. When those patients are asked why they’re quitting statins, the answer for about 60% of them is ‘muscle symptoms’—principally myalgia. As noted in a recent review: “muscle-related side effects, the fear of side effects, and perception of side effects are among the common reasons patients often report for declining to start and for discontinuing statin therapy.”
This might seem like a lot of patients are stopping statins for a legitimate reason. But they’re mostly quitting because of the nocebo effect: they believe statins have side effects that they largely do not. Several experiments speak to this fact.
ASCOT-LLA: This study had two phases, one blinded and randomized, and another that was unblinded and unrandomized. In the RCT phase, participants took either atorvastatin or a placebo and reported on side effects. The critical, muscle-related side effects occurred at a rate of 2.03% per year in the statin group and 2.00% per year in the placebo group—virtually identical rates. But when blinding was removed, participants who were on statins reported an annual rate of muscle-related problems of 1.26% versus 1.00% for those who weren’t on statins. It took knowing about the statins for them to generate muscular problems!2
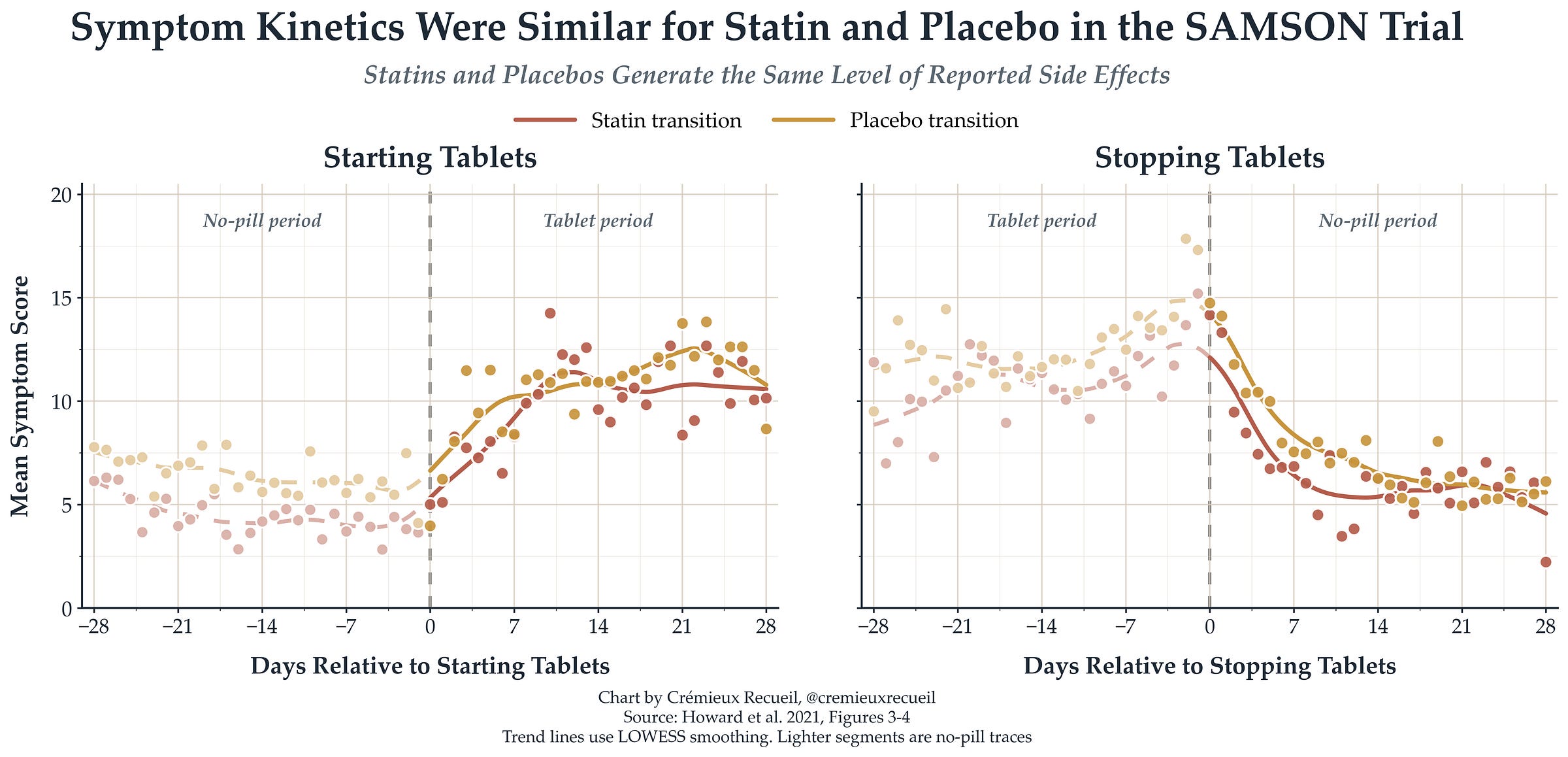
SAMSON: This study took people who had previously stopped statins because of side effects and cycled them through atorvastatin, placebo, and no-tablet months to see how their side effect reports progressed. When people went on statins or placebo pills, they started reporting more symptoms, and they did so to similar degrees. When they came off either pill, they recovered to similar degrees, at the same rate. It was taking any pill that mattered to patients, not taking statins specifically. Placebo was enough.
StatinWISE: This study had patients who had previously stopped or were considering stopping statins because of muscle symptoms alternate between two-month periods of taking either statins or placebo. Participants rated their muscular symptoms at the end of each treatment period and there was no difference between periods taking statins or placebo. The difference was a minuscule -0.11 points on the 0-10-point visual analogue scale, nonsignificantly favoring statins. An alternative measure is withdrawals from the study, and by that measure, there were no significant differences: 9% of participants dropped during statin periods and 7% dropped during placebo period—a nonsignificant result favoring placebo.
Two-thirds of participants who completed the study intended to stick with treatment.
The nocebo-eliciting trials are clear, as are the results from a review of rechallenge trials in which doctors try to get people back on statins after they come off. That review showed no significant differences in global and myalgia symptom ratings, and 36% of the rechallenged participants assigned to statins developed an intolerance versus 26% on placebo. It is also commonly-stated consensus that over 90% of patients who stopped treatment due to side effects are able to successfully continue on a statin when they’re rechallenged, though much of this is based on observational studies, where that percentage visibly do that, albeit without randomization.
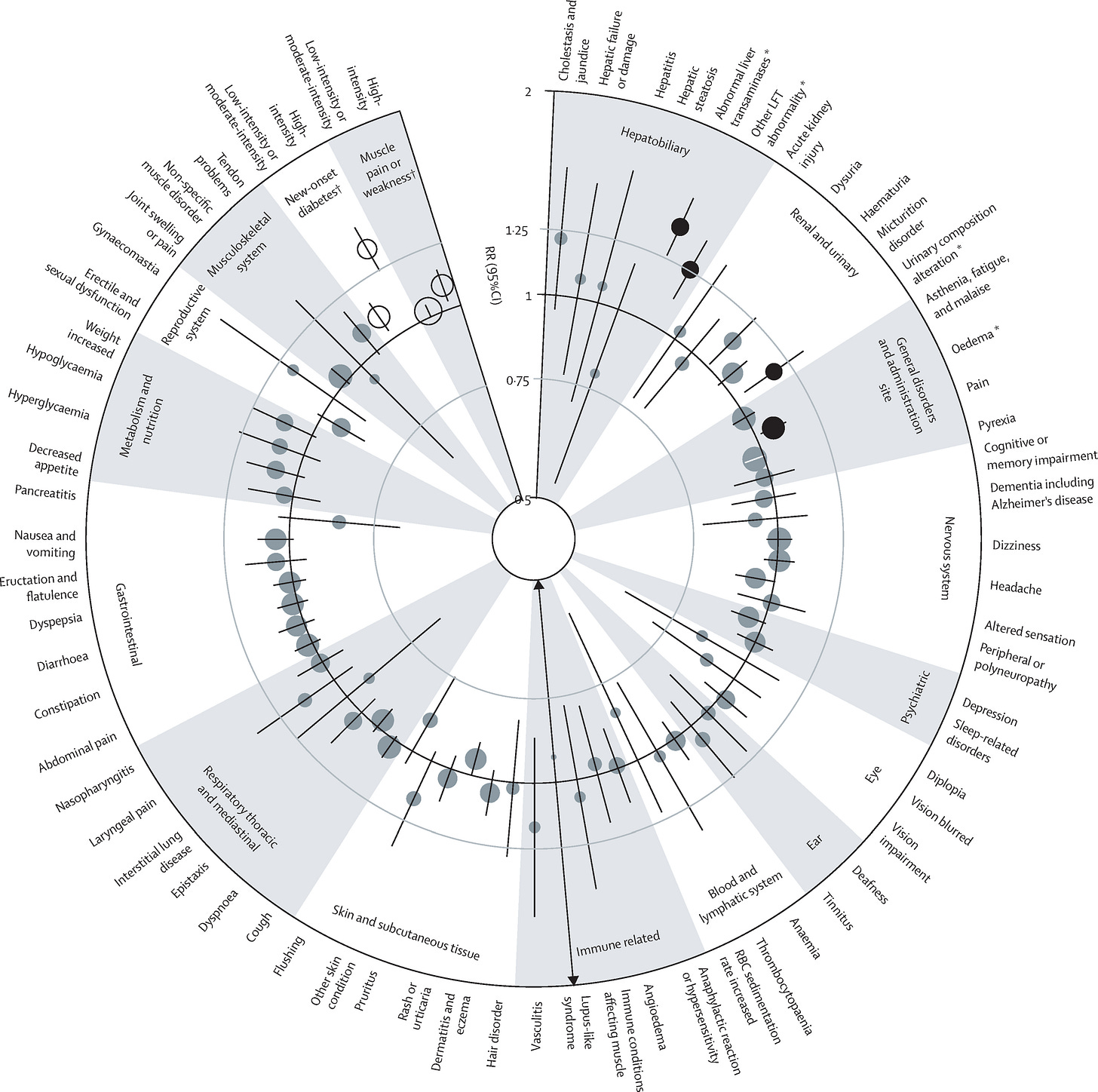
All of this is no surprise when placebo-controlled trials show support for very few of the side effects listed on statin labels, and what they do show is universally meager. A recent review of nineteen trials with 123,940 participants in it included this diagram:
If you can’t tell, there is not a single clinically-meaningful effect size pictured in that ring. Moreover, almost every result is null, and given that they almost all cover a very limited range, they are almost all very precise nulls.3 The largest positive effect—meaning one in which statins lead to side effects—is equivalent to about r = 0.10 or d = 0.19. It’s practically nothing, and, as the authors have noted elsewhere: “any theoretical adverse effects of statins on cardiovascular risk that might arise from [their side effects] (or, indeed, from any other mechanism) are already accounted for in the overall reduction in cardiovascular risk that is seen with statin therapy”.4
I’m not going to keep harping on statins. They are a great example where there’s a real issue, but its scale has been so overstated and overhyped that it’s helped to generate nocebo effects in real-world patient populations. Celiac is like this in the sense that it’s a real and serious issue, and it seems to have given birth, by a feat of logic, to non-celiac gluten sensitivity, which is also more strongly nocebo- than substance-driven.
Examples like these speak to the power of belief, suggestion, or whatever you’d like to call it. But nocebo effects aren’t the only way belief matters in health.
People can also be prompted to think they’ve been exposed to things because that have become salient. For example, after a series of high-profile court cases about talc and ovarian cancer hit the news, people started remembering that they used talc. What an odd thing to suddenly remember, and such convenient timing, too!5
Ultimately, what I’m getting at is that the old adage that patients always lie has a kernel of truth to it. But the lies aren’t always to doctors per se, and they’re not always malicious or conscious. In fact, what I’m really saying is: I think the person that patients are the most likely to lie to is none other than themselves.6
For comparison, people who genetically have the effects of taking statins are also better off.
Other side effects are worth noting. In the randomized phase, their rates just didn’t differ in any meaningful way: muscle-related side effect rates were comparable, as noted above; erectile dysfunction rates were basically identical (1.86% per year for statin takers vs. 2.14% for placebo takers); sleep impairment rates were actually somewhat lower in the statin group (1.00% vs. 1.46%); cognitive impairment rates were identical (0.20% vs. 0.22%); and all other side-effects were similarly matched, except for an excess of renal and urinary side-effects among statin users (1.87% vs. 1.51%).
Another nice finding is that the effects match what we see in genetic studies via Mendelian Randomization. For example, the very small effect on weight is observed with Mendelian Randomization. This makes sense because LDL-affecting genes mirror LDL-affecting drugs in their effects, so differences are rarely observed and are practically never meaningful.
For muscular symptoms specifically, the chart shows the risk elevation is minor, and studies specifically covering this have concluded the same. In that review of trials, 27.1% of those on statins and 26.6% of those on placebo reported muscle pain or weakness—a statistically significant but not clinically meaningful difference on average.
I’ve already mentioned it, but want to repeat since it’s worth keeping in mind: for those patients who actually have issues, they can switch to another statin that doesn’t elicit said issue (this works for large portions of patients) or take ezetimibe, PCSK9 inhibitors, etc.
And, on a more tangential subject, people can be prompted to believe that certain things are healthy or unhealthy, so they start consuming or avoiding them, making their cross-sectional associations unreliable. To learn more about that, see:
I would like, for once, for people suffering from psychosomatic illnesses to not grossly misinterpret statements like this.